
P.S./CHEMISTRY
P.S./CHEMISTRY
The University of the State of New York
REGENTS HIGH SCHOOL EXAMINATION
PHYSICAL SETTING
CHEMISTRY
Friday, January 24, 2020 — 9:15 a.m. to 12:15 p.m., only
The possession or use of any communications device is strictly prohibited when
taking this examination. If you have or use any communications device, no matter how
briefl y, your examination will be invalidated and no score will be calculated for you.
This is a test of your knowledge of chemistry. Use that knowledge to answer all
questions in this examination. Some questions may require the use of the 2011 Edition
Reference Tables for Physical Setting/Chemistry. You are to answer all questions in all
parts of this examination according to the directions provided in this examination
booklet.
A separate answer sheet for Part A and Part B–1 has been provided to you. Follow
the instructions from the proctor for completing the student information on your
answer sheet. Record your answers to the Part A and Part B–1 multiple-choice
questions on this separate answer sheet. Record your answers for the questions in
Part B–2 and Part C in your separate answer booklet. Be sure to fi ll in the heading on
the front of your answer booklet.
All answers in your answer booklet should be written in pen, except for graphs and
drawings, which should be done in pencil. You may use scrap paper to work out the
answers to the questions, but be sure to record all your answers on your separate
answer sheet or in your answer booklet as directed.
When you have completed the examination, you must sign the statement printed
on your separate answer sheet, indicating that you had no unlawful knowledge of the
questions or answers prior to the examination and that you have neither given nor
received assistance in answering any of the questions during the examination. Your
answer sheet and answer booklet cannot be accepted if you fail to sign this declaration.
Notice. . .
A four-function or scientifi c calculator and a copy of the 2011 Edition Reference Tables for
Physical Setting/Chemistry must be available for you to use while taking this examination.
DO NOT OPEN THIS EXAMINATION BOOKLET UNTIL THE SIGNAL IS GIVEN.

P.S. Chem.–Jan. ’20 [2]
1 Which statement describes a concept included in
the wave-mechanical model of the atom?
(1) Protons, neutrons, and electrons are located
in the nucleus.
(2) Electrons orbit the nucleus in shells at fi xed
distances.
(3) Atoms are hard, indivisible spheres.
(4) Electrons are located in regions called
orbitals.
2 As an electron in an atom moves from a higher
energy state to a lower energy state, the atom
(1) becomes a negative ion
(2) becomes a positive ion
(3) releases energy
(4) absorbs energy
3 Two atoms that are different isotopes of the same
element have
(1) the same number of protons and the same
number of neutrons
(2) the same number of protons but a different
number of neutrons
(3) a different number of protons but the same
number of neutrons
(4) a different number of protons and a different
number of neutrons
4 The element in Group 14, Period 3, of the
Periodic Table is classifi ed as a
(1) metal (3) metalloid
(2) noble gas (4) nonmetal
5 Which element has chemical properties that are
most similar to potassium?
(1) calcium (3) nitrogen
(2) cesium (4) sulfur
6 Which element requires the least amount of
energy to remove the most loosely held electron
from a gaseous atom in the ground state?
(1) Na (3) P
(2) Ar (4) Cl
7 Which terms identify two different categories of
compounds?
(1) covalent and molecular
(2) covalent and empirical
(3) ionic and molecular
(4) ionic and empirical
8 Which statement describes the energy and
bonding changes as two atoms of fl uorine become
a molecule of fl uorine?
(1) Energy is absorbed as a bond is broken.
(2) Energy is absorbed as a bond is formed.
(3) Energy is released as a bond is broken.
(4) Energy is released as a bond is formed.
9 Which part of a calcium atom in the ground state
is represented by the dots in its Lewis electron-
dot diagram?
(1) the electrons in the fi rst shell
(2) the electrons in the fourth shell
(3) the protons in the nucleus
(4) the neutrons in the nucleus
10 Based on Table S, an atom of which element
has the strongest attraction for electrons in a
chemical bond?
(1) aluminum (3) magnesium
(2) chlorine (4) sulfur
Part A
Answer all questions in this part.
Directions (1–30): For each statement or question, record on your separate answer sheet the number of the
word or expression that, of those given, best completes the statement or answers the question. Some questions
may require the use of the 2011 Edition Reference Tables for Physical Setting/Chemistry.
P.S. Chem.–Jan. ’20 [3] [OVER]
11 Which substance can not be broken down by
chemical means?
(1) aluminum
(2) ammonia
(3) aluminum oxide
(4) ammonium chloride
12 Which statement describes the particles of an
ideal gas, based on the kinetic molecular theory?
(1) There are attractive forces between the
particles.
(2) The particles move in circular paths.
(3) The collisions between the particles reduce
the total energy of the gas.
(4) The volume of the gas particles is negligible
compared with the total volume of the gas.
13 What is the amount of heat released by 1.00 gram
of liquid water at 0°C when it changes to 1.00 gram
of ice at 0°C?
(1) 4.18 J (3) 334 J
(2) 273 J (4) 2260 J
14 Which term identifi es a type of intermolecular
force?
(1) covalent bonding (3) ionic bonding
(2) hydrogen bonding (4) metallic bonding
15 Which statement describes a reaction at
equilibrium?
(1) The mass of the products must equal the
mass of the reactants.
(2) The entropy of the reactants must equal the
entropy of the products.
(3) The rate of formation of the products must
equal the rate of formation of the reactants.
(4) The number of moles of the reactants must
equal the number of moles of the products.
16 Entropy is a measure of
(1) accuracy
(2) precision
(3) the disorder of a system
(4) the attraction of a nucleus for an electron
17 Systems in nature tend to undergo changes
toward
(1) lower energy and less randomness
(2) higher energy and less randomness
(3) lower energy and greater randomness
(4) higher energy and greater randomness
18 Which organic prefi x is matched with the number
of carbon atoms that it represents?
(1) hept-, 7 (3) pent-, 3
(2) non-, 8 (4) prop-, 4
19 Which terms represent two types of organic
reactions?
(1) sublimation and deposition
(2) sublimation and fermentation
(3) saponifi cation and deposition
(4) saponifi cation and fermentation
20 Given the organic functional group:
C
O
H
Which class of organic compounds has molecules
with this functional group?
(1) aldehydes (3) ketones
(2) esters (4) organic acids
21 Which particles are transferred during a redox
reaction?
(1) atoms (3) neutrons
(2) electrons (4) positrons
22 Which process can be represented by a half-
reaction equation?
(1) distillation (3) sublimation
(2) oxidation (4) vaporization
23 Which form of energy is converted to electrical
energy in a voltaic cell?
(1) chemical (3) nuclear
(2) mechanical (4) thermal
P.S. Chem.–Jan. ’20 [4]
24 Which compound is an Arrhenius base?
(1) HCl (3) Ca(OH)
2
(2) H
3
PO
4
(4) CH
3
COOH
25 In a neutralization reaction, an aqueous solution
of an Arrhenius acid reacts with an aqueous
solution of an Arrhenius base to produce
(1) an ether and water
(2) an ether and an alcohol
(3) a salt and water
(4) a salt and an alcohol
26 According to one acid-base theory, a base is an
(1) H
2
acceptor (3) H
⫹
acceptor
(2) H
2
donor (4) H
⫹
donor
27 Based on Table N, uranium-238 and uranium-235
have different
(1) decay modes
(2) half-lives
(3) numbers of protons
(4) numbers of electrons
28 A change in the nucleus of an atom that converts
the atom from one element to another element is
called
(1) oxidation-reduction
(2) single replacement
(3) substitution
(4) transmutation
29 Which radioactive emission has the greatest
penetrating power, but the least ionizing power?
(1) alpha particle (3) gamma ray
(2) beta particle (4) positron
30 Which statement describes a benefi t of using
fi ssion reactions?
(1) Radioactive waste must be stored for long
periods of time.
(2) Nuclear fuel consists of stable isotopes.
(3) Gamma radiation is produced.
(4) Large amounts of energy are produced per
mole of reactant.

P.S. Chem.–Jan. ’20 [5] [OVER]
31 Given the table representing the subatomic
particles in four different atoms:
Atom
Number of
Protons
Number of
Neutrons
Number of
Electrons
A444
E575
G676
J121212
Which atom has a mass of 12 u?
(1) A (3) G
(2) E (4) J
32 Which electron confi guration could represent
the electrons in a sodium atom in an excited
state?
(1) 2-8 (3) 2-7-1
(2) 2-8-1 (4) 2-7-2
33 What is the number of valence electrons in a
nitrogen atom in the ground state?
(1) 5 (3) 7
(2) 2 (4) 14
34 Graphite and diamond are both solid forms of
the element carbon. Which statement explains
the different properties of these two forms of
carbon?
(1) Diamond has ionic bonding and graphite has
metallic bonding.
(2) Diamond has metallic bonding and graphite
has ionic bonding.
(3) Diamond has a different crystal structure
from graphite.
(4) Diamond has carbon atoms with more
valence electrons than graphite.
35 A measured value for the atomic radius
of platinum atoms was determined to be
143 picometers. Based on Table S, what is the
percent error of this measured value?
(1) 0.10% (3) 10.%
(2) 9.1% (4) 13%
36 What is the chemical formula for sodium oxalate?
(1) NaO (3) NaC
2
O
4
(2) Na
2
O (4) Na
2
C
2
O
4
37 Given the formula of a compound:
C
H
H
C
H
C
H
H
H
What is the molecular formula for this compound?
(1) CH (3) CH
3
(2) CH
2
(4) C
3
H
6
38 Which equation represents conservation of
charge?
(1) I
⫺
⫹ 2e
⫺
→ I
2
(3) Br
2
→ 2Br
⫺
⫹ 2e
⫺
(2) 2I
⫺
→ I
2
⫹ 2e
⫺
(4) Br ⫹ 2e
⫺
→ Br
⫺
39 Which equation represents a single replacement
reaction?
(1) 2Al(s) ⫹ 3Cl
2
(g) → 2AlCl
3
(s)
(2) 2Al(s) ⫹ 6HCl(aq) → 2AlCl
3
(aq) ⫹ 3H
2
(g)
(3) 2AlCl
3
(s) → 2Al(s) ⫹ 3Cl
2
(g)
(4) AlCl
3
(aq) ⫹ 3KOH(aq) → Al(OH)
3
(s) ⫹ 3KCl(aq)
40 The bond between which two atoms is most
polar?
(1) C–O (3) H–O
(2) F–F (4) N–H
Part B–1
Answer all questions in this part.
Directions (31–50): For each statement or question, record on your separate answer sheet the number of the
word or expression that, of those given, best completes the statement or answers the question. Some questions
may require the use of the 2011 Edition Reference Tables for Physical Setting/Chemistry.
P.S. Chem.–Jan. ’20 [6]
41 The table below shows the volume and
temperature of four different gas samples at
100. kPa.
Gas
Sample
Volume
(L)
Temperature
(°C)
helium 1 25
neon 2 50.
argon 1 25
krypton 2 25
Which two gas samples contain equal numbers
of atoms?
(1) helium and neon
(2) helium and argon
(3) neon and argon
(4) neon and krypton
42 Given the equation representing a solution
equilibrium:
BaSO
4
(s)
H
2
O
Ba
2⫹
(aq) ⫹ SO
4
2⫺
(aq)
What occurs when Na
2
SO
4
(s) is added to this
system, increasing the concentration of SO
4
2⫺
(aq)?
(1) The equilibrium shifts to the left, and the
concentration of Ba
2⫹
(aq) decreases.
(2) The equilibrium shifts to the left, and the
concentration of Ba
2⫹
(aq) increases.
(3) The equilibrium shifts to the right, and the
concentration of Ba
2⫹
(aq) decreases.
(4) The equilibrium shifts to the right, and the
concentration of Ba
2⫹
(aq) increases.
43 Given the formula for a compound:
C
H
H
HC
H
H
C
H
H
C
O
N
H
H
What is a chemical name for the compound?
(1) 1-butanamine (3) butanamide
(2) 1-butanol (4) butanoic acid
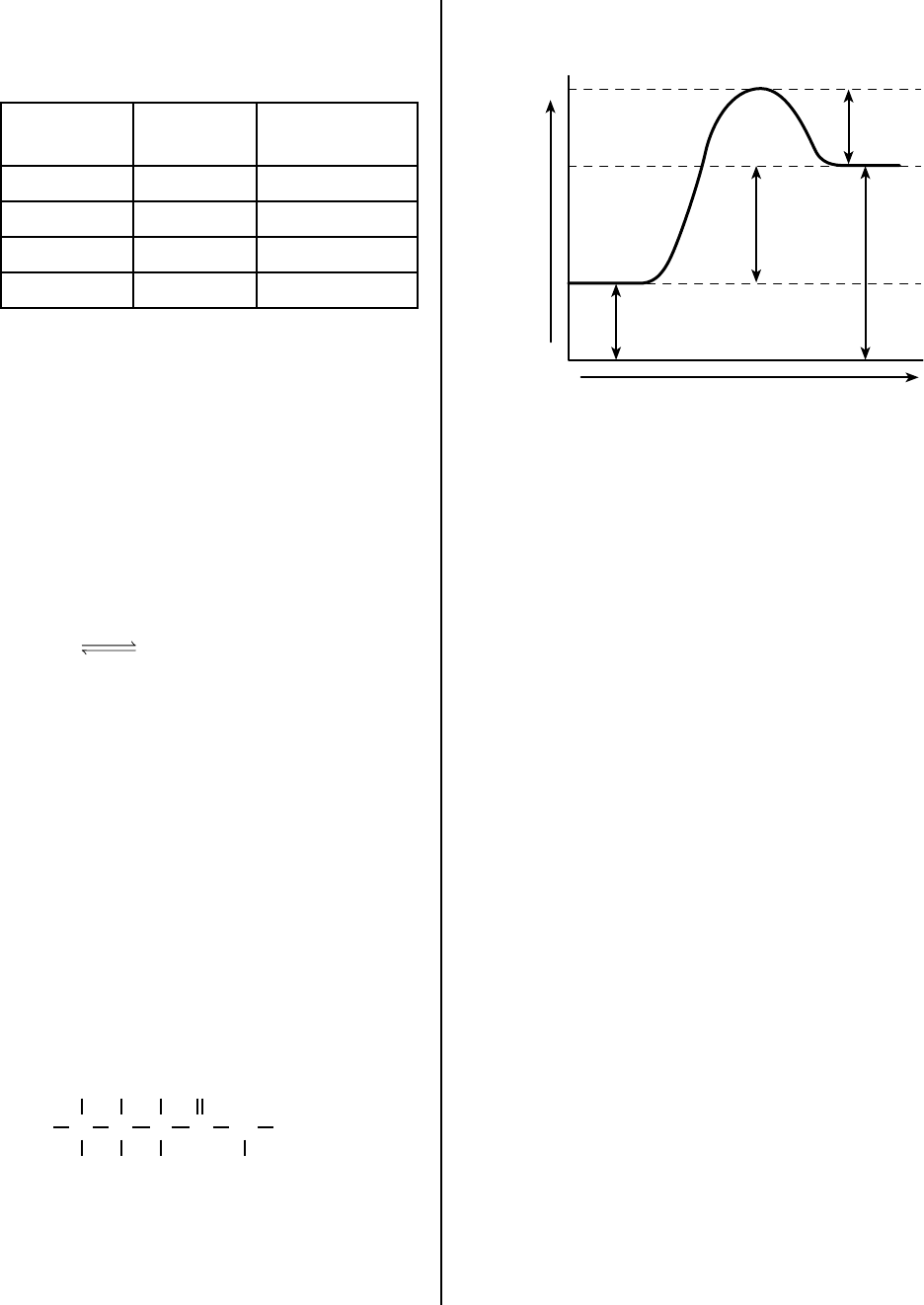
44 Given the potential energy diagram representing
a reaction:
Reaction Coordinate
Potential Energy
1
2
3
4
Which numbered interval represents the heat of
reaction?
(1) 1 (3) 3
(2) 2 (4) 4
45 When comparing voltaic cells to electrolytic cells,
oxidation occurs at the
(1) anode in both types of cells
(2) cathode in both types of cells
(3) anode in voltaic cells, only
(4) cathode in voltaic cells, only
46 Based on Table J, which metal is more active
than tin, but less active than zinc?
(1) Ag (3) Cs
(2) Cr (4) Mn
47 In a titration, 10.0 mL of 0.0750 M HCl(aq) is
exactly neutralized by 30.0 mL of KOH(aq)
of unknown concentration. What is the
concentration of the KOH(aq) solution?
(1) 0.0250 M (3) 0.225 M
(2) 0.0750 M (4) 0.333 M
48 Which emission causes the atomic number of a
nuclide to decrease by 2 and its mass number to
decrease by 4?
(1) an alpha particle (3) gamma radiation
(2) a beta particle (4) a positron

P.S. Chem.–Jan. ’20 [7] [OVER]
49 The diagram below represents the bright-line spectra of four elements and a bright-line spectrum produced
by an unidentifi ed element.
Bright-Line Spectra
500 600 700
Wavelength (nm)
Element L
500 600 700
Element M
Element X
Element Z
Unidentified Element
What is the unidentifi ed element?
(1) L (3) X
(2) M (4) Z
50 Given two equations representing reactions:
Equation 1:
235
92
U ⫹
1
0
n →
141
56
Ba ⫹
92
36
Kr ⫹ 3
1
0
n
Equation 2:
1
1
H⫹
2
1
H →
3
2
He
Which type of reaction is represented by each of these equations?
(1) Both equations represent fi ssion.
(2) Both equations represent fusion.
(3) Equation 1 represents fi ssion and equation 2 represents fusion.
(4) Equation 1 represents fusion and equation 2 represents fi ssion.
P.S. Chem.–Jan. ’20 [8]
Base your answers to questions 51 through 53 on the information below and on your knowledge of chemistry.
The four naturally occurring isotopes of sulfur are S-32, S-33, S-34, and S-36. The table
below shows the atomic mass and percent natural abundance for these isotopes.
Naturally Occurring Isotopes of Sulfur
Isotope
Atomic
Mass
(u)
Natural
Abundance
(%)
S-32 31.972 94.99
S-33 32.971 0.75
S-34 33.968 4.25
S-36 35.967 0.01
51 State both the number of protons and the number of neutrons in an S-33 atom. [1]
52 In the space in your answer booklet, show a numerical setup for calculating the atomic
mass of sulfur. [
1]
53 Compare the energy of an electron in the third shell of a sulfur atom to the energy of
an electron in the fi rst shell of the same atom. [
1]
Part B–2
Answer all questions in this part.
Directions (51-65): Record your answers in the spaces provided in your answer booklet. Some questions may
require the use of the 2011 Edition Reference Tables for Physical Setting/Chemistry.
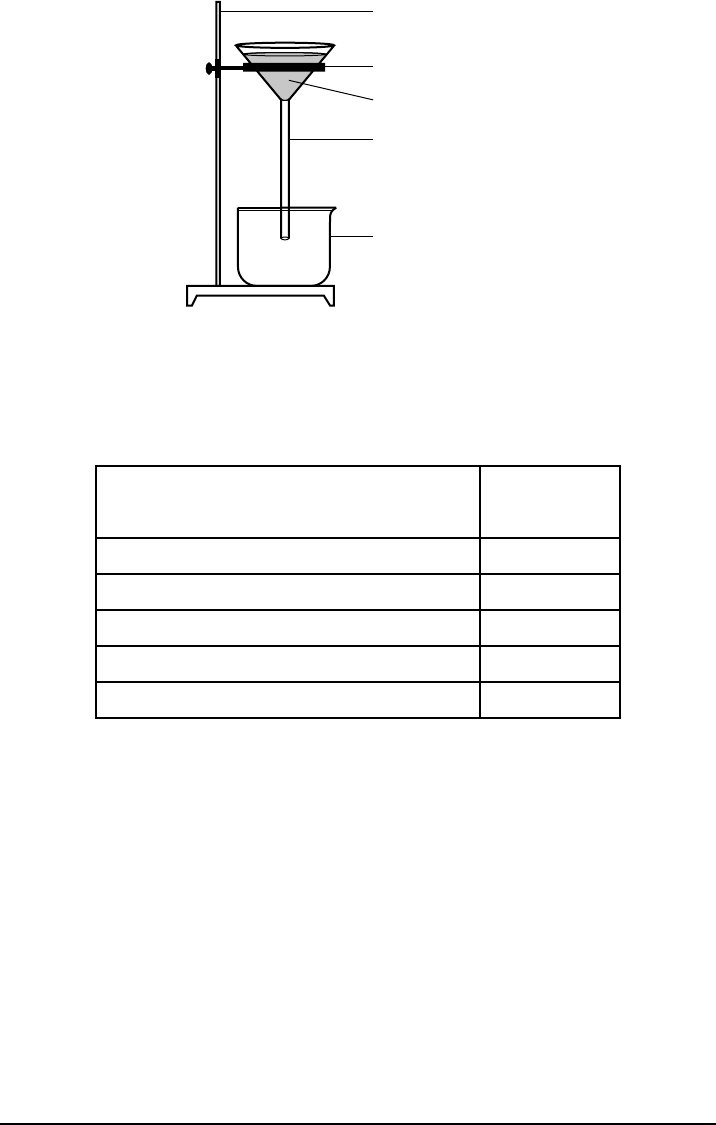
P.S. Chem.–Jan. ’20 [9] [OVER]
Base your answers to questions 54 through 57 on the information below and on your knowledge of chemistry.
During a laboratory activity, appropriate safety equipment is used and safety procedures
are followed. A student separates a sample of rock salt that has two components; NaCl(s)
and small insoluble rock particles. First, the student thoroughly stirs the sample of rock salt
into a sample of water in a fl ask. The mixture in the fl ask is fi ltered using the lab apparatus
shown below.
Ring stand
Iron ring
Filter paper
Glass funnel
Beaker
The water is evaporated from the beaker. The fi lter paper and its contents are dried.
The data collected by the student are shown in the table below.
Rock Salt Separation Lab Data
Object or Material
Mass
(g)
rock salt sample 16.4
fi lter paper 1.6
clean empty beaker 224.2
fi lter paper with dry rock particles 2.2
beaker with dry NaCl(s) 240.0
54 State evidence, other than mass, from the information given that the components of rock
salt have different properties. [
1]
55 Explain, in terms of particle size, why the rock particles are trapped by the fi lter
paper. [
1]
56 State the number of signifi cant fi gures in the mass of the beaker with dry NaCl(s). [
1]
57 Show a numerical setup for calculating the percent by mass of NaCl in the rock salt
sample. [
1]
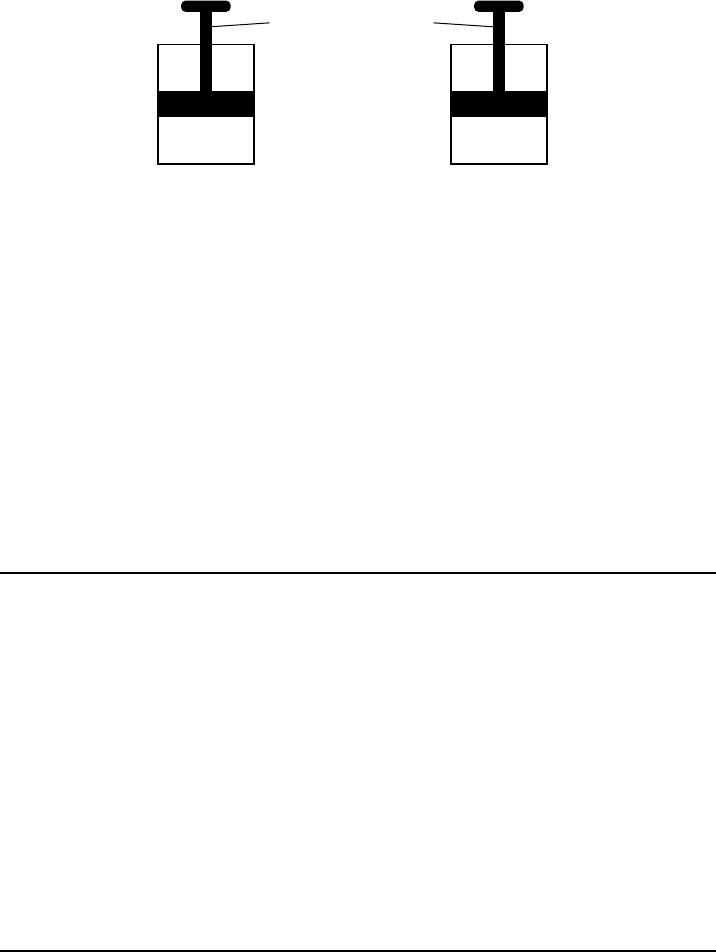
P.S. Chem.–Jan. ’20 [10]
Base your answers to questions 58 through 61 on the information below and on your knowledge of chemistry.
Cylinder A and cylinder B are sealed, rigid cylinders with movable pistons. Each
cylinder contains 500. milliliters of a gas sample at 101.3 kPa and 298 K. Cylinder A contains
H
2
(g) and cylinder B contains N
2
(g). The diagrams below represent these two cylinders.
Cylinder A Cylinder B
Movable piston
H
2
(g) N
2
(g)
58 Compare the mass of the gas in cylinder A to the mass of the gas in cylinder B. [1]
59 State a change in temperature and a change in pressure that will cause the gas in
cylinder A to behave more like an ideal gas. [
1]
60 Explain, in terms of collisions between gas molecules and the walls of the container,
why pushing the movable piston farther into cylinder B at constant temperature would
increase the pressure of the N
2
gas. [1]
61 Show a numerical setup for calculating the volume of the gas in cylinder B at STP. [
1]
Base your answers to questions 62 and 63 on the information below and on your knowledge of chemistry.
The electrical conductivity of three aqueous solutions was tested at room temperature.
A 0.1 M HCl(aq) solution conducted, but a 6.0 M HCl(aq) solution was a better conductor.
A 0.1 M C
6
H
12
O
6
(aq) solution was also tested. During this laboratory activity, appropriate
safety equipment was used and safety procedures were followed.
62 State, in terms of the concentration of ions, why the 6.0 M HCl(aq) is a better
conductor of electricity than the 0.1 M HCl(aq). [
1]
63 Identify the element in C
6
H
12
O
6
that allows it to be classifi ed as an organic compound. [1]

P.S. Chem.–Jan. ’20 [11] [OVER]
Base your answers to questions 64 and 65 on the information below and on your knowledge of chemistry.
Phosphorus-30 and phosphorus-32 are radioisotopes. Phosphorus-30 decays by positron
emission.
64 Complete the equation in your answer booklet for the decay of phosphorus-30 by writing
a notation for the missing product. [
1]
65 Based on Table N, determine the time required for an original 100.-milligram sample
of P-32 to decay until only 25 milligrams of the sample remain unchanged. [
1]

P.S. Chem.–Jan. ’20 [12]
Base your answers to questions 66 through 69 on the information below and on your knowledge of chemistry.
Sir William Ramsey is one scientist credited with identifying the noble gas argon.
Sir Ramsey separated nitrogen gas from the air and reacted it with an excess of magnesium,
producing solid magnesium nitride. However, a small sample of an unreactive gas remained
with a density different from the density of the nitrogen gas. Sir Ramsey identifi ed the
unreactive gas as argon and later went on to discover neon, krypton, and xenon.
66 Compare the chemical reactivities of nitrogen gas and argon gas based on Sir Ramsey’s
experiment using magnesium. [
1]
67 Compare the density of nitrogen gas to the density of argon gas when both gases are at
298 K and 101.3 kPa. [
1]
68 State, in terms of valence electrons, why the noble gases that Sir Ramsey discovered have
similar chemical properties. [
1]
69 State the trend, at standard pressure, of the boiling points of these noble gases, as they
are considered in order of increasing atomic number. [
1]
Base your answers to questions 70 through 72 on the information below and on your knowledge of chemistry.
A sample of normal rainwater has a pH value of 5.6 due to dissolved carbon dioxide gas
from the atmosphere. Acid rain is formed when other gases, such as sulfur dioxide, dissolve
in rainwater, which can result in lake water with a pH value of 4.6. The equation below
represents the reaction of water with SO
2
(g).
H
2
O( ) ⫹ SO
2
(g) → H
2
SO
3
(aq)
70 State how many times greater the hydronium ion concentration in the lake water is than
the hydronium concentration in the sample of normal rainwater. [
1]
71 State the color of methyl orange in a sample of normal rainwater. [
1]
72 Based on Table G, describe what happens to the solubility of SO
2
(g) as the temperature
increases from 10.°C to 30.°C at standard pressure. [1]
Part C
Answer all questions in this part.
Directions (66-85): Record your answers in the spaces provided in your answer booklet. Some questions may
require the use of the 2011 Edition Reference Tables for Physical Setting/Chemistry.

P.S. Chem.–Jan. ’20 [13] [OVER]
Base your answers to questions 73 through 77 on the information below and on your knowledge of chemistry.
A metal worker uses a cutting torch that operates by reacting acetylene gas, C
2
H
2
(g),
with oxygen gas, O
2
(g), as shown in the unbalanced equation below.
C
2
H
2
(g) ⫹ O
2
(g) → CO
2
(g) ⫹ H
2
O(g) ⫹ heat
73 Write the empirical formula for acetylene. [
1]
74 In your answer booklet, use the key to draw a particle model diagram to represent the
phase of the O
2
(g). Your response must include at least six molecules. [1]
75 Balance the equation in your answer booklet for the reaction of acetylene and oxygen,
using the smallest whole-number coeffi cients. [
1]
76 Determine the mass of 25 moles of acetylene (gram-formula mass = 26 g/mol). [
1]
77 Explain, in terms of bonding, why the hydrocarbon gas used in the cutting torch is
classifi ed as an alkyne. [
1]
P.S. Chem.–Jan. ’20 [14]
Base your answers to questions 78 through 82 on the information below and on your knowledge of chemistry.
Water, H
2
O, and hexane, C
6
H
14
, are commonly used as laboratory solvents because they
have different physical properties and are able to dissolve different types of solutes. Some
physical properties of water and hexane are listed on the table below.
Physical Properties of H
2
O and C
6
H
14
Solvent
Boiling Point
(°C)
Melting Point
(°C)
Vapor Pressure at 69°C
(kPa)
H
2
O 100. 0. ?
C
6
H
14
69 –95 101.3
78 Compare the thermal energy of a 10.-gram sample of water at 25°C to the thermal
energy of a 1000.-gram sample of water at 25°C. [1]
79 State what happens to the potential energy of the molecules in a solid sample of hexane
at –95°C as heat is added until the hexane is completely melted. [
1]
80 Determine the vapor pressure of water at 69°C. [
1]
81 Explain, in terms of the molecular polarity, why hexane is nearly insoluble in water. [
1]
82 Explain, in terms of molecular formulas and structural formulas, why
2,2-dimethylbutane is an isomer of hexane. [
1]
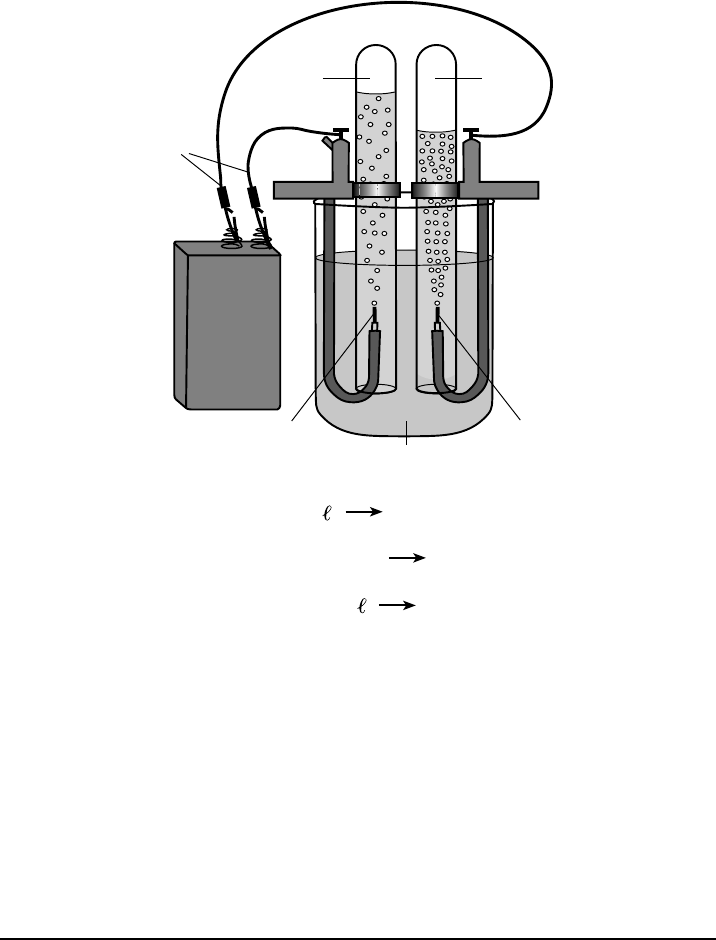
P.S. Chem.–Jan. ’20 [15] [OVER]
Base your answers to questions 83 through 85 on the information below and on your knowledge of chemistry.
In a laboratory investigation, a student constructs an electrochemical cell to decompose
water, as represented in the diagram below. The water in the electrochemical cell contains a
small amount of dissolved sodium sulfate, to increase conductivity. The three equations
represent the reaction in each test tube and the overall reaction. During this laboratory activity,
appropriate safety equipment is used and safety procedures are followed.
+–
Battery
H
2
Test Tube: 2H
+
(aq) + 2e
–
H
2
(g)
O
2
Test Tube: 2H
2
O( ) O
2
(g) + 4H
+
(aq) + 4e
–
Overall Reaction: 2H
2
O( ) O
2
(g) + 2H
2
(g)
Cathode
Anode
Wires
(g)
2
O H (g)
2
Water
83 State the change in oxidation number that occurs for oxygen in the overall reaction. [1]
84 Compare the number of electrons lost by oxygen to the number of electrons gained by
hydrogen in the overall reaction. [
1]
85 Determine the number of moles of hydrogen gas produced when 0.0004 mole of
oxygen gas is produced in the cell by the overall reaction. [
1]
P.S./CHEMISTRY
P.S./CHEMISTRY
Printed on Recycled Paper

The University of the State of New York
REGENTS HIGH SCHOOL EXAMINATION
PHYSICAL SETTING
CHEMISTRY
Friday, January 24, 2020 — 9:15 a.m. to 12:15 p.m., only
ANSWER BOOKLET
Student . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
Teacher. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
School . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . Grade . . . . . . . . .
Record your answers for Part B–2 and Part C in this booklet.
51 Protons: _____________
Neutrons: _____________
52
53
Part B–2

54
55
56 _____________
57
P.S./Chem. Answer Booklet–Jan. ’20 [2]

58
59 Temperature:
Pressure:
60
61
P.S./Chem. Answer Booklet–Jan. ’20 [3] [OVER]

62
63
64
30
15
P →
0
⫹1
e ⫹ _____________
65 _____________ d
P.S./Chem. Answer Booklet–Jan. ’20 [4]

P.S./Chem. Answer Booklet–Jan. ’20 [5] [OVER]
66
67
68
69
70
71
72
Part C

P.S./Chem. Answer Booklet–Jan. ’20 [6]
73
74
75 _________ C
2
H
2
(g) ⫹ _________ O
2
(g) → _________ CO
2
(g) ⫹ _________ H
2
O(g) ⫹ heat
76 g
77
78
79
Key
= an oxygen molecule

80 kPa
81
82
83 From ____________ to ____________
84
85 mol
P.S./Chem. Answer Booklet–Jan. ’20 [7]

P.S./Chem. Answer Booklet–Jan. ’20
P.S./CHEMISTRY
P.S./CHEMISTRY
Printed on Recycled Paper
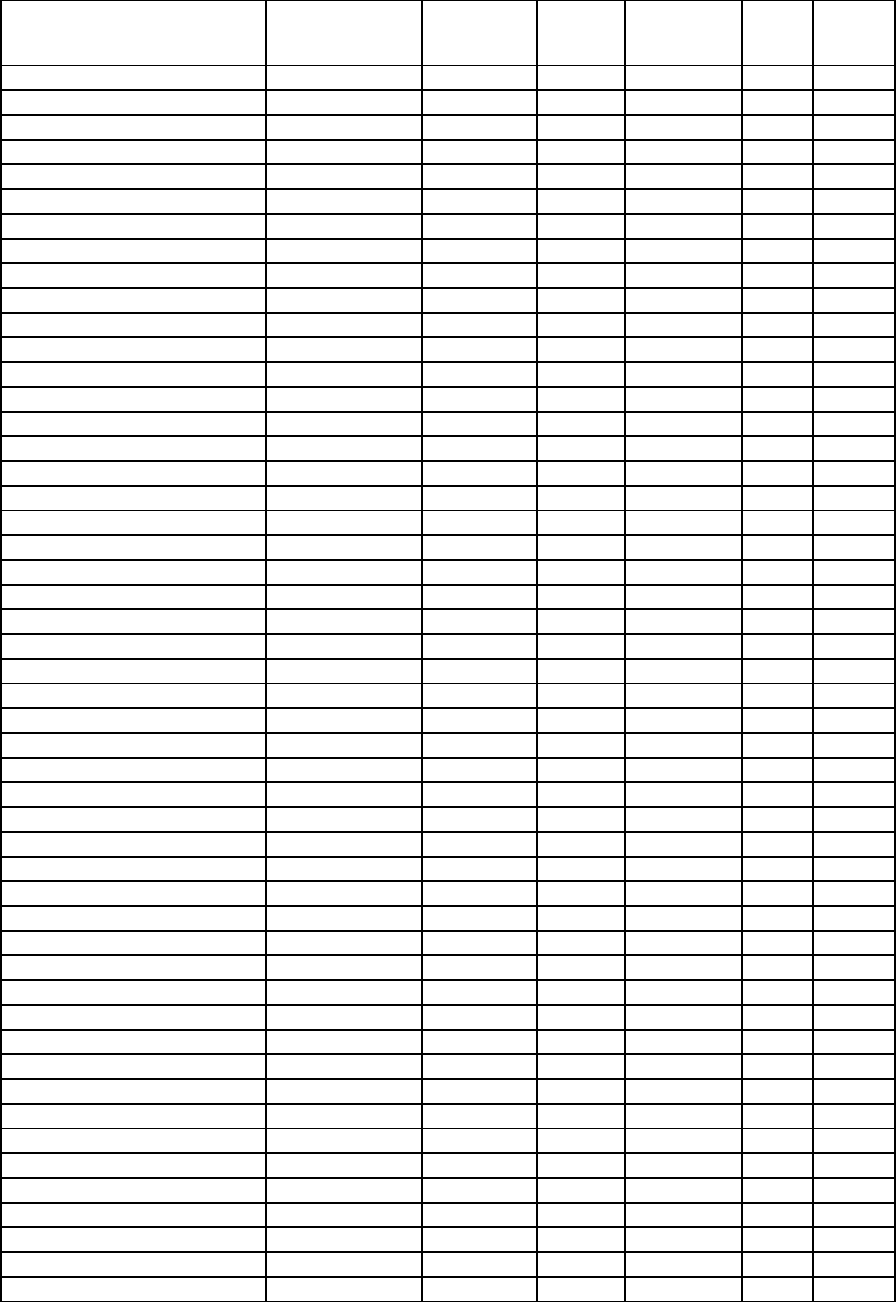
Examination Date
Question
Number
Scoring
Key
Question
Type
Credit Weight
Physical Setting/Chemistry
January '20
1 4
MC 1 1
Physical Setting/Chemistry
January '20
2 3
MC
1 1
Physical Setting/Chemistry
January '20
3 2
MC 1 1
Physical Setting/Chemistry
January '20
4 3
MC 1 1
Physical Setting/Chemistry
January '20
5 2
MC
1 1
Physical Setting/Chemistry
January '20
6 1
MC 1 1
Physical Setting/Chemistry
January '20
7 3
MC
1 1
Physical Setting/Chemistry
January '20
8 4
MC
1 1
Physical Setting/Chemistry
January '20
9 2
MC 1 1
Physical Setting/Chemistry
January '20
10 2
MC
1 1
Physical Setting/Chemistry
January '20
11 1
MC 1 1
Physical Setting/Chemistry
January '20
12 4
MC 1 1
Physical Setting/Chemistry
January '20
13
3
MC 1 1
Physical Setting/Chemistry
January '20
14 2
MC
1 1
Physical Setting/Chemistry
January '20
15
3
MC
1 1
Physical Setting/Chemistry
January '20
16 3
MC 1 1
Physical Setting/Chemistry
January '20
17 3
MC
1 1
Physical Setting/Chemistry
January '20
18 1
MC 1 1
Physical Setting/Chemistry
January '20
19 4
MC
1 1
Physical Setting/Chemistry
January '20
20 1
MC
1 1
Physical Setting/Chemistry
January '20
21 2
MC 1 1
Physical Setting/Chemistry
January '20
22 2
MC 1 1
Physical Setting/Chemistry
January '20
23 1
MC
1 1
Physical Setting/Chemistry
January '20
24 3
MC 1 1
Physical Setting/Chemistry
January '20
25 3
MC 1 1
Physical Setting/Chemistry
January '20
26 3
MC
1 1
Physical Setting/Chemistry
January '20
27 2
MC 1 1
Physical Setting/Chemistry
January '20
28 4
MC
1 1
Physical Setting/Chemistry
January '20
29 3
MC
1 1
Physical Setting/Chemistry
January '20
30 4
MC 1 1
Physical Setting/Chemistry
January '20
31 2
MC
1 1
Physical Setting/Chemistry
January '20
32 4
MC 1 1
Physical Setting/Chemistry
January '20
33 1
MC
1 1
Physical Setting/Chemistry
January '20
34 3
MC 1 1
Physical Setting/Chemistry
January '20
35 3
MC
1 1
Physical Setting/Chemistry
January '20
36 4
MC
1 1
Physical Setting/Chemistry
January '20
37 4
MC 1 1
Physical Setting/Chemistry
January '20
38 2
MC
1 1
Physical Setting/Chemistry
January '20
39 2
MC 1 1
Physical Setting/Chemistry
January '20
40 3
MC
1 1
Physical Setting/Chemistry
January '20
41 2
MC 1 1
Physical Setting/Chemistry
January '20
42 1
MC
1 1
Physical Setting/Chemistry
January '20
43 3
MC
1 1
Physical Setting/Chemistry
January '20
44 2
MC 1 1
Physical Setting/Chemistry
January '20
45 1
MC
1 1
Physical Setting/Chemistry
January '20
46 2
MC 1 1
Physical Setting/Chemistry
January '20
47 1
MC 1 1
Physical Setting/Chemistry
January '20
48 1
MC
1 1
Physical Setting/Chemistry
January '20
49 1
MC 1 1
Physical Setting/Chemistry
January '20
50 3
MC
1 1
The State Education Department / The University of the State of New York
Regents Examination in Physical Setting/Chemistry – January 2020
Scoring Key: Parts A and B-1 (Multiple-Choice Questions)
P.S./Chemistry Scoring Key 1 of 2

Examination Date
Question
Number
Scoring
Key
Question
Type
Credit Weight
Physical Setting/Chemistry
January '20
51 -
CR
1 1
Physical Setting/Chemistry
January '20
52 -
CR 1 1
Physical Setting/Chemistry
January '20
53 -
CR 1 1
Physical Setting/Chemistry
January '20
54 -
CR
1 1
Physical Setting/Chemistry
January '20
55 -
CR 1 1
Physical Setting/Chemistry
January '20
56 -
CR
1 1
Physical Setting/Chemistry
January '20
57 -
CR 1 1
Physical Setting/Chemistry
January '20
58 -
CR
1 1
Physical Setting/Chemistry
January '20
59 -
CR 1 1
Physical Setting/Chemistry
January '20
60 -
CR
1 1
Physical Setting/Chemistry
January '20
61 -
CR 1 1
Physical Setting/Chemistry
January '20
62
-
CR
1 1
Physical Setting/Chemistry
January '20
63 -
CR 1 1
Physical Setting/Chemistry
January '20
64 -
CR
1 1
Physical Setting/Chemistry
January '20
65 -
CR 1 1
Physical Setting/Chemistry
January '20
66 -
CR 1 1
Physical Setting/Chemistry
January '20
67 -
CR
1 1
Physical Setting/Chemistry
January '20
68 -
CR 1 1
Physical Setting/Chemistry
January '20
69 -
CR 1 1
Physical Setting/Chemistry
January '20
70 -
CR
1 1
Physical Setting/Chemistry
January '20
71 -
CR 1 1
Physical Setting/Chemistry
January '20
72 -
CR
1 1
Physical Setting/Chemistry
January '20
73 -
CR 1 1
Physical Setting/Chemistry
January '20
74 -
CR 1 1
Physical Setting/Chemistry
January '20
75 -
CR
1 1
Physical Setting/Chemistry
January '20
76 -
CR
1 1
Physical Setting/Chemistry
January '20
77 -
CR
1 1
Physical Setting/Chemistry
January '20
78 -
CR 1 1
Physical Setting/Chemistry
January '20
79 -
CR
1 1
Physical Setting/Chemistry
January '20
80 -
CR
1 1
Physical Setting/Chemistry
January '20
81
-
CR 1 1
Physical Setting/Chemistry
January '20
82
-
CR
1 1
Physical Setting/Chemistry
January '20
83 -
CR 1 1
Physical Setting/Chemistry
January '20
84 -
CR
1 1
Physical Setting/Chemistry
January '20
85 -
CR 1 1
Key
MC = Multiple-choice question
The chart for determining students' final examination scores for the January 2020 Regents
Examination in Physical Setting/Chemistry will be posted on the Department’s web site at
http://www.p12.nysed.gov/assessment/
on the day of the examination. Conversion charts provided
for the previous administrations of the Physical Setting/Chemistry examination must NOT be used to
determine students’ final scores for this administration.
Scoring Key: Parts B-2 and C (Constructed-Response Questions)
CR = Constructed-response question
P.S./Chemistry Scoring Key 2 of 2

FOR TEACHERS ONLY
The University of the State of New York
REGENTS HIGH SCHOOL EXAMINATION
PHYSICAL SETTING/CHEMISTRY
Friday, January 24, 2020 — 9:15 a.m. to 12:15 p.m., only
RATING GUIDE
Directions to the Teacher:
Refer to the directions on page 2 before rating student papers.
Updated information regarding the rating of this examination may be posted on the New York
State Education Department’s web site during the rating period. Check this web site
at: http://www.p12.nysed.gov/assessment/
and select the link “Scoring Information” for any
recently posted information regarding this examination. This site should be checked before the
rating process for this examination begins and several times throughout the Regents Examination
period.

P.S./Chem. Rating Guide–Jan. ’20 [2]
Directions to the Teacher
Follow the procedures below for scoring student answer papers for the Regents Examination in
Physical Setting/Chemistry. Additional information about scoring is provided in the publication
Information Booklet for Scoring Regents Examinations in the Sciences.
At least two science teachers must participate in the scoring of the Part B–2 and Part C open-ended
questions on a student’s paper. Each of these teachers should be responsible for scoring a selected number
of the open-ended questions on each answer paper. No one teacher is to score more than approximately
one-half of the open-ended questions on a student’s answer paper. Teachers may not score their own
students’ answer papers.
Students’ responses must be scored strictly according to the Rating Guide. For open-ended questions,
credit may be allowed for responses other than those given in the rating guide if the response is a scientifically
accurate answer to the question and demonstrates adequate knowledge, as indicated by the examples in the
rating guide. Do not attempt to correct the student’s work by making insertions or changes of any kind. On
the student’s separate answer sheet, for each question, record the number of credits earned and the
teacher’s assigned rater/scorer letter.
Fractional credit is not allowed. Only whole-number credit may be given for a response. If the student
gives more than one answer to a question, only the first answer should be rated. Units need not be given
when the wording of the questions allows such omissions.
For hand scoring, raters should enter the scores earned in the appropriate boxes printed on the
separate answer sheet. Next, the rater should add these scores and enter the total in the box labeled
“Total Raw Score.” Then the student’s raw score should be converted to a scale score by using the
conversion chart that will be posted on the Department’s web site at: http://www.p12.nysed.gov/assessment/
on Friday, January 24, 2020. The student’s scale score should be entered in the box labeled “Scale Score”
on the student’s answer sheet. The scale score is the student’s final examination score.
Schools are not permitted to rescore any of the open-ended questions on this exam after each
question has been rated once, regardless of the final exam score. Schools are required to ensure
that the raw scores have been added correctly and that the resulting scale score has been
determined accurately.
Because scale scores corresponding to raw scores in the conversion chart may change from one
administration to another, it is crucial that, for each administration, the conversion chart provided for that
administration be used to determine the student’s final score.

P.S./Chem. Rating Guide–Jan. ’20 [3]
51 [1] Allow 1 credit for 16 protons and 17 neutrons.
52 [1] Allow 1 credit. Acceptable responses include, but are not limited to:
(31.972 u)(0.9499) ⫹ (32.971 u)(0.0075) ⫹ (33.968 u)(0.0425) ⫹ (35.967 u)(0.0001)
31.972(94.99) ⫹ 32.971(0.75) ⫹ 33.968(4.25) ⫹ (35.967)(0.01)
100
4.25%(33.968) ⫹ .75%(32.971) ⫹ 94.99%(31.972) ⫹ .01%(35.967)
Note: Do not allow credit for a numerical setup using mass numbers rather than isotopic masses.
53 [1] Allow 1 credit. Acceptable responses include, but are not limited to:
The energy of an electron in the third shell is higher than the energy of an electron in the first
shell.
The third shell electron has higher energy.
The electron in the first shell has less.
Note: The student response must address energy of electrons, not just shells.
54 [1] Allow 1 credit. Acceptable responses include, but are not limited to:
The NaCl is soluble in water, and the rock particles are insoluble.
The mixture can be separated by filtration.
55 [1] Allow 1 credit. Acceptable responses include, but are not limited to:
The particles of the rock are much larger than the openings in the filter paper.
The rock particles are too big to pass through the paper.
56 [1] Allow 1 credit for 4 or four.
Part B–2
Allow a total of 15 credits for this part. The student must answer all questions in this part.

P.S./Chem. Rating Guide–Jan. ’20 [4]
57 [1] Allow 1 credit. Acceptable responses include, but are not limited to:
(240.0 g ⫺ 224.2 g) ⫻ 100
16.4 g
(100)(15.8)
16.4
16.4 ⫺ 0.6 ⫻ 100
16.4
15.8
____
⫽
x
____
16.4 100
Note: Do not allow credit if the fraction is not multiplied by 100.
58 [1] Allow 1 credit. Acceptable responses include, but are not limited to:
The gas in cylinder A has a smaller mass than the mass of the gas in cylinder B.
The nitrogen gas has more mass.
The H
2
(g) in cylinder A has less mass.
59 [1] Allow 1 credit. Acceptable responses include, but are not limited to:
Temperature: increase
Pressure: decrease
Temperature: above 25°C
Pressure: below 1.00 atm
Temperature: any temperature above 298 K
Pressure: any pressure below 101.3 kPa
60 [1] Allow 1 credit. Acceptable responses include, but are not limited to:
Moving the piston farther into the cylinder would increase the number of collisions per unit
area between the nitrogen molecules and the inside walls of the cylinder, creating greater
pressure.
There would be more collisions, causing a higher pressure.

61 [1] Allow 1 credit. Acceptable responses include, but are not limited to:
(101.3 kPa)(500. mL)
⫽
(101.3 kPa)(V
2
)
298 K 273 K
(500 mL)(273 K)
298 K
(500)(273)
298
62 [1] Allow 1 credit. Acceptable responses include, but are not limited to:
There is a greater concentration of ions present in the 6.0 M HCl(aq) than in the
0.1 M HCl(aq).
The 6.0 M HCl(aq) has a higher concentration of ions.
Note: Do not allow credit for “more ions” because it is not in terms of concentrations of ions.
63 [1] Allow 1 credit for C or carbon.
64 [1] Allow 1 credit. Acceptable responses include, but are not limited to:
30
14
Si
30
Si
silicon-30
Si-30
65 [1] Allow 1 credit for 28.56 d. Significant figures do not need to be shown.
P.S./Chem. Rating Guide–Jan. ’20 [5]

P.S./Chem. Rating Guide–Jan. ’20 [6]
66 [1] Allow 1 credit. Acceptable responses include, but are not limited to:
The Ar in the sample did not react, and the nitrogen did.
Magnesium reacted with the nitrogen gas, and the argon gas did not react.
Nitrogen is more reactive.
67 [1] Allow 1 credit. Acceptable responses include, but are not limited to:
The density of nitrogen gas is less than the density of argon gas.
Argon is more dense.
Nitrogen gas has a density of 0.001145 g/cm
3
, which is less than the density of argon.
68 [1] Allow 1 credit. Acceptable responses include, but are not limited to:
Their atoms have the same number of valence electrons.
These elements have similar chemical properties because their atoms have valence electron
shells with a complete octet.
Their outermost shells have 8 e
⫺
.
They have a full outermost shell of electrons.
69 [1] Allow 1 credit. Acceptable responses include, but are not limited to:
As the atomic number of these elements increases, their boiling points increase.
Boiling point goes up as atomic number gets larger.
Part C
Allow a total of 20 credits for this part. The student must answer all questions in this part.
70 [1] Allow 1 credit. Acceptable responses include, but are not limited to:
10
ten
tenfold
10 times
71 [1] Allow 1 credit for yellow.
72 [1] Allow 1 credit. Acceptable responses include, but are not limited to:
As the water temperature increases, the solubility of sulfur dioxide decreases.
The solubility of SO
2
decreases.
The SO
2
(g) becomes less soluble.
73 [1] Allow 1 credit for CH or HC.
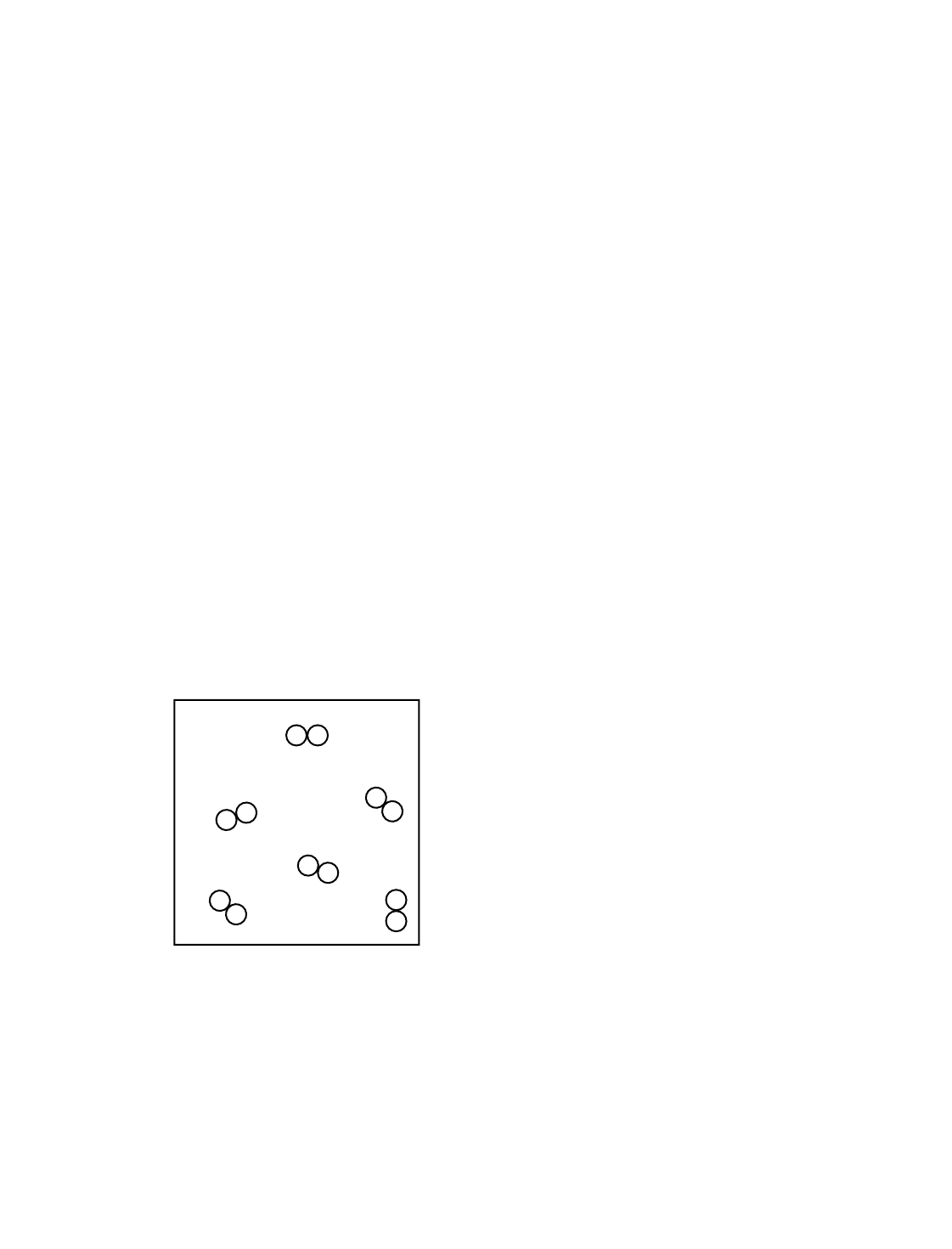
74 [1] Allow 1 credit for a diagram with at least six diatomic molecules drawn to represent the gas phase of
the sample.
Example of a 1-credit response:
75 [1] Allow 1 credit for
2542
_________ C
2
H
2
(g) ⫹ _________ O
2
(g) → _________ CO
2
(g) ⫹ _________ H
2
O(g) ⫹ heat
76 [1] Allow 1 credit for 650 g or any value from 650 g to 651 g, inclusive.
P.S./Chem. Rating Guide–Jan. ’20 [7]

77 [1] Allow 1 credit. Acceptable responses include, but are not limited to:
Each molecule has a triple carbon-to-carbon bond, C⫽
⫺
C.
The two C atoms share 6 electrons.
Each molecule has a triple bond.
Alkynes have a C⫽
⫺
C.
78 [1] Allow 1 credit. Acceptable responses include, but are not limited to:
The thermal energy is greater for the 1000 g sample of water.
The smaller sample has less thermal energy.
79 [1] Allow 1 credit. Acceptable responses include, but are not limited to:
The potential energy increases.
P.E. goes up.
80 [1] Allow 1 credit for any value from 28 kPa to 30. kPa, inclusive.
81 [1] Allow 1 credit. Acceptable responses include, but are not limited to:
Hexane molecules are nonpolar, and water molecules are polar.
Water and hexane have different molecular polarities.
82 [1] Allow 1 credit. Acceptable responses include, but are not limited to:
The hexane and the 2,2-dimethylhexane have the same molecular formula but have different
structural formulas.
Both molecules have the same number of C atoms and the same number of H atoms but have
a different arrangement of atoms.
Both compounds are C
6
H
14
, but have different structures.
P.S./Chem. Rating Guide–Jan. ’20 [8]

83 [1] Allow 1 credit. Acceptable responses include, but are not limited to:
From ⫺2 to 0
From 2⫺ to 0
From negative two to zero
Note: Do not allow credit for 2 without a minus sign (⫺).
84 [1] Allow 1 credit. Acceptable responses include, but are not limited to:
The number of electrons lost by oxygen is equal to the number of electrons gained by hydrogen.
The same number of electrons is lost and gained.
equal
same
85 [1] Allow 1 credit for 0.0008 mol or 8 × 10
⫺4
mol. Significant figures do not need to be shown.
P.S./Chem. Rating Guide–Jan. ’20 [9]

P.S./Chem. Rating Guide–Jan. ’20 [10]
The Chart for Determining the Final Examination Score for the January 2020
Regents Examination in Physical Setting/Chemistry will be posted on
the Department’s web site at: http://www.p12.nysed.gov/assessment/
on
Friday, January 24, 2020. Conversion charts provided for previous administra-
tions of the Regents Examination in Physical Setting/Chemistry must NOT be
used to determine students’ final scores for this administration.
Regents Examination in Physical Setting/Chemistry
January 2020
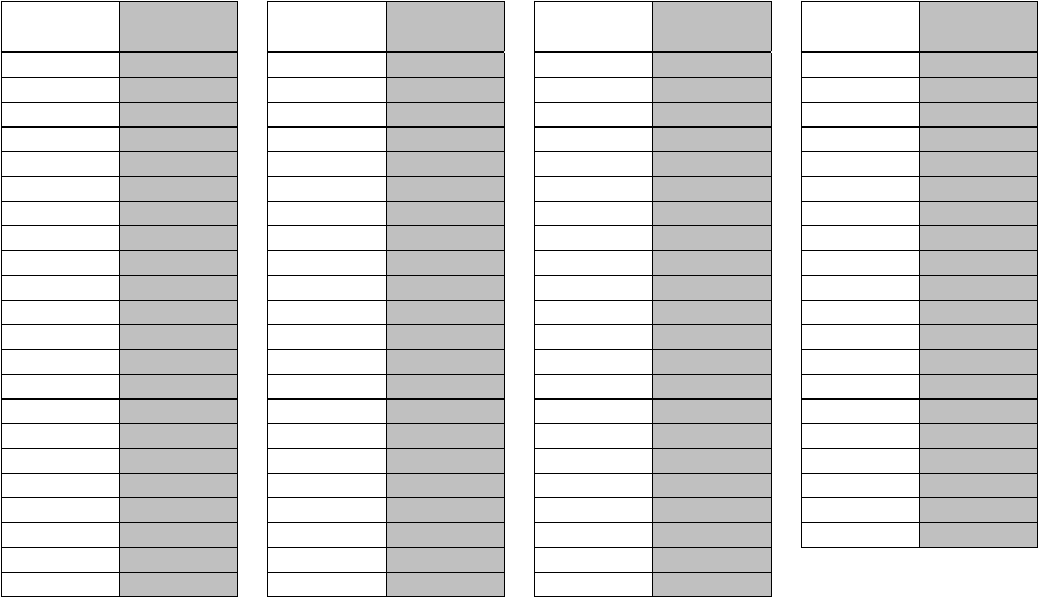
Chart for Converting Total Test Raw Scores to
Final Examination Scores (Scale Scores)
Online Submission of Teacher Evaluations of the Test to the Department
Suggestions and feedback from teachers provide an important contribution to the test
development process. The Department provides an online evaluation form for State
assessments. It contains spaces for teachers to respond to several specific questions and to
make suggestions. Instructions for completing the evaluation form are as follows:
1. Go to http://www.forms2.nysed.gov/emsc/osa/exameval/reexameval.cfm
.
2. Select the test title.
3. Complete the required demographic fields.
4. Complete each evaluation question and provide comments in the space provided.
5. Click the SUBMIT button at the bottom of the page to submit the completed form.

P.S./Chem. Rating Guide–Jan. ’20 [11]
0DSWR&RUH&XUULFXOXP
January 2020 Physical Setting/Chemistry
Question Numbers
Key Ideas/Performance Indicators Part A Part B Part C
Standard 1
Math Key Idea 1 52, 56, 57, 61
Math Key Idea 2 41, 48 70, 79
Math Key Idea 3 35, 36, 37, 40, 47,
51, 64, 65
73, 75, 76, 83, 85
Science Inquiry Key Idea 1 34, 45, 46, 50, 51,
53, 54, 55, 58, 59,
60, 62, 63, 64
66, 67, 68, 69, 77,
81, 82, 84
Science Inquiry Key Idea 2
Science Inquiry Key Idea 3 31, 36, 37, 39, 42,
43, 46, 48, 49, 50,
63
66, 67, 73, 77, 80,
82, 83
Engineering Design Key Idea 1
Standard 2
Key Idea 1 54
Key Idea 2
Key Idea 3
Standard 6
Key Idea 1
Key Idea 2 35 74
Key Idea 3 70
Key Idea 4
Key Idea 5 72, 80
Standard 7
Key Idea 1
Key Idea 2
Standard 4 Process Skills
Key Idea 3 32, 33, 38, 39, 42,
45, 46, 47, 49, 51,
52, 59,61
69, 71, 72, 73, 74,
75, 76, 85
Key Idea 4 44, 50, 64, 65 78, 79
Key idea 5 40, 62
Standard 4
Key Idea 3 1, 2, 3, 4, 5, 6, 11,
12, 15, 16, 17, 18,
19, 20, 21, 22, 23,
24, 25, 26, 29
31, 32, 33, 35, 36,
37, 38, 39, 41, 42,
43, 45, 46, 47, 48,
49, 51, 52, 53, 54,
55, 56, 57, 58, 59,
60, 61, 62, 63
66, 67, 68, 69,70,
71, 72, 73, 74, 75,
76, 77, 82, 83, 84,
85
Key Idea 4 13, 27, 30 44, 50, 64, 65 78, 79
Key Idea 5 7, 8, 9, 10, 14, 28 34, 40 80, 81
Reference Tables
2011 Edition 4, 5, 6, 9, 10, 13,
18, 20, 24, 27, 29
32, 33, 35, 36, 40,
43, 46, 47, 48, 50,
51, 53, 57, 58, 59,
61, 64, 65
66, 67, 68, 69, 71,
72, 76, 77, 80, 82,
83
Raw Scale Raw Scale Raw Scale Raw Scale
Score Score Score Score Score Score Score Score
85
100
63
75
41
59
19
38
84
98
62
74
40
58
18
37
83
97
61
73
39
58
17
36
82
95
60
72
38
57
16
34
81
94
59
72
37
56
15
33
80
92
58
71
36
56
14
31
79
91
57
70
35
55
13
29
78
90
56
69
34
54
12
28
77
88
55
69
33
53
11
26
76
87
54
68
32
52
10
24
75
86
53
67
31
51
9
22
74
85
52
67
30
50
8
20
73
84
51
66
29
49
7
18
72
83
50
66
28
48
6
16
71
82
49
65
27
47
5
14
70
81
48
64
26
46
4
11
69
80
47
63
25
45
3
9
68
79
46
63
24
44
2
6
67
78
45
62
23
43
1
3
66
77
44
61
22
42
0
0
65
76
43
60
21
41
64
75
42
60
20
40
Schools are not permitted to rescore any of the open-ended questions on this exam after each question has
been rated once, regardless of the final exam score. Schools are required to ensure that the raw scores have
been added correctly and that the resulting scale score has been determined accurately.
Because scale scores corresponding to raw scores in the conversion chart change from one administration to
another, it is crucial that for each administration the conversion chart provided for that administration be used to
determine the student’s final score. The chart above is usable only for this administration of the Regents Examination
in Physical Setting/Chemistry.
The State Education Department / The University of the State of New York
Regents Examination in Physical Setting/Chemistry – January 2020
Chart for Converting Total Test Raw Scores to Final Examination Scores (Scale Scores)
To determine the student’s final examination score, find the student’s total test raw score in the column labeled “Raw
Score” and then locate the scale score that corresponds to that raw score. The scale score is the student’s final
examination score. Enter this score in the space labeled “Scale Score” on the student’s answer sheet.
P.S./Chemistry 1 of 1
